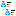First photos of the atom
Oct 6 2009 · Random
Last month physicists at the Kharkov Institute for Physics and Technology photographed the electron cloud of a single carbon atom. The images are the first real photographs of the electrons of a single atom, showing the s and p orbitals of a carbon atom.

Quantum mechanics states that an electron doesn’t exist as a single point, but spreads around the nucleus in a cloud known as an orbital. The soft blue spheres and split clouds seen in the images show two arrangements of the electrons in their orbitals in a carbon atom. The structures verify illustrations seen in thousands of chemistry books because they match established quantum mechanical predictions.
… and as for how it was done…
To create these images, the researchers used a field-emission electron microscope, or FEEM. They placed a rigid chain of carbon atoms, just tens of atoms long, in a vacuum chamber and streamed 425 volts through the sample. The atom at the tip of the chain emitted electrons onto a surrounding phosphor screen, rendering an image of the electron cloud around the nucleus
…
The sharper a sample’s pointed tip inside the vacuum chamber, the greater the resolution of the final image on the screen said Igor Mikhailovskij, one of the paper’s authors. In the last year, physicists learned to manipulate carbon atoms into chains. With the tip of the sample now just a single atom wide, the microscope was able to resolve the electron’s orbitals.